Your Cart is Empty
In October 2018, Purifas® conducted a clinical trial on the difference in bacterial transfer from the client to the therapy bed with and without the use of the FaceShield™.
As part of the study, we followed a stringent three step cleaning protocol to ensure the bed was sterile before we commenced treatment to guarantee accurate data collection. A swab of the bed was taken to ensure the bed was sterile.
The first aspect of the study, physiotherapy treatment without the FaceShield™, replicates the current lack of protection around the face hole, with a swab taken after five patients to determine bacterial transfer (Figure 1).
This process was repeated with the FaceShield™ to assess the reduction in bacterial transfer as a result of using the FaceShield™ (Figure 2).
Images of the agar plates comparing the levels of bacterial growth are below. It is evident from the images that there is a significant reduction in bacterial transfer with the use of the FaceShield™.

|
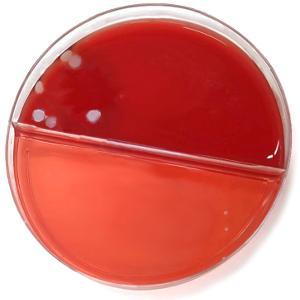 |
|
Figure 1: No FaceShield™ used |
Figure 2: FaceShield™ used |
As the FaceShield™ filters bacteria in both directions, if the bed was infected with pathogens prior to commencing therapy, our FaceShield would also protect the consumer as it reduces their exposure to these microbes.
Helping therapists thrive, not just survive running a hands-on practice can be rewarding but it also comes with burnout, business stress and physical strain. That’s where The Health Leader Co. steps in. With decades of experience across clinical excellence, business mentoring and marketing strategy, Elicia Crook, Christine Knox and James Crook are empowering therapists to build sustainable, fulfilling careers. From advanced training to mindset shifts and practical tools, this trio is helping practitioners love their practice again. Explore the full blog to learn how The Health Leader Co. can support your growth, protect your wellbeing and help you build a practice you truly love.
Immerse yourself in a world of unparalleled comfort and hygiene with Purifas. This vivid narrative illustrates an exceptional spa experience, showcasing the BodyShield, Dream Topper and FaceShield’s superior cleanliness, comfort and luxury over traditional towels. It highlights their silent, aesthetic appeal, setting a new standard for health and wellness treatments, leaving the author eager to visit only Purifas-equipped clinics and salons.